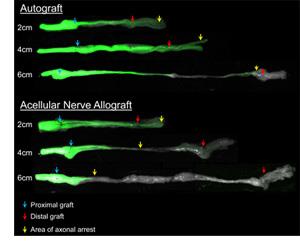
Images of nerve isografts and acellular nerve scaffolds used to repair a nerve defect in Thy1-GFP rats. Axon growth is seen in green. Severe peripheral nerve injuries can result in a defect generated within the nerve. This results in loss of function as signals from the neurons through axons cannot be transmitted to the end-organs (skin and/or muscle). In some instances, direct nerve repair may not be possible (either end-to-end or via nerve transfer). Therefore, a “bridge” is needed to facilitate axon regeneration across the gap or defect. A nerve autograft, or nerve tissue taken from the patient, remains the standard of care to bridge the gap, as it can robustly facilitate axon regeneration across the defect. During this procedure, the patient’s native nerves, usually “expendable” superficial sensory nerves, are harvested for transfer to the surgical site. Autografts have disadvantages for the patient including donor site morbidity from the tissue harvest and increased operating time and risk. Due to the disadvantages of autografts, there has been a long-standing interest in developing bioengineered substitutes or alternatives for autografts.
We determined that both autografts and alternatives to autografts, such as nerve guidance conduits or tissue-engineered devices, have more limitations to treat nerve injuries than previously thought. As the length of the graft increases, regeneration and functional recovery decreases due to the graft. As a possible cause, the lab has been studying glial and support cells comprising nerve that regulate new tissue formation as well as guide axons to regenerate in a functional manner. Using acellular nerve as a model nerve graft alterative, these studies have revealed that the cell repopulation of acellular nerve scaffolds differs between short and long grafts. While cell repopulation leading to functional recovery is adequate for short acellular nerve, these recent studies have demonstrated that long acellular nerve are repopulated with a cellular population imbalance, consisting of increased populations of stromal cells as well as Schwann cells expressing markers of senescence, compared to short scaffolds. We think these cumulative changes, especially the accumulation and persistence of senescent cells, represent a “barrier” to axon regeneration.